 ATP binding to the NBD induces the SBD open conformation by separating the Base and Lid subdomains, a conformation of low affinity for the binding of client proteins. The SBD can adopt two conformations: open and closed ( Figure 2B). These hydrophobic sequences are usually buried in the protein core and their accessibility signals a misfolded conformation. The SBD recognizes a motif present in most proteins on average every 30 to 40 residues this short five-residue motif is enriched in hydrophobic amino acids and is flanked by regions with predominantly positively charged residues 13. NBD subdomains are referred to as IB, IIB, IA, and IIA. ( B) Hsp70 atomic structures in the closed (PDB 2KHO) and open (PDB 4JNE) conformations. For simplicity, the numbering below the scheme shows only the first amino acid of each domain. Numeration is according to heat shock 70-kDa protein 1B Homo sapiens NP_005337.2. NBD: nucleotide-binding domain, aa 1-381 L: linker, aa 382-397 SBD Base: substrate-binding domain Base, aa 398-505 SBD Lid: substrate-binding domain Lid, aa 506-605 CTD: C-terminal domain, aa 606-641. The length of the segments is in scale with amino acid length. ( A) Scheme of heat shock protein 70 (Hsp70) domain organization. Heat shock protein 70 (Hsp70)-domain organization and atomic structures of functional conformations. Here, we will discuss some of the challenges that Hsp70 and Hsp70-based complexes offer for structural biologists.įigure 2. Obtaining high-resolution structural information of the different Hsp70-based protein complexes will contribute considerably to understanding the molecular mechanisms in protein homeostasis. 
However, this information is critical for interfering with or modulating these pathways with therapeutic purposes in pathological conditions. Several reports have already confirmed this expectation CMA appears to involve exclusively the constitutive Hsc70 isoform 8, human Hsp70 and Hsc70 differ considerably in their disaggregation potentials 9 and, surprisingly, while Hsc70 protects microtubule-associated protein tau (MAPT) from degradation, Hsp70 accelerates its clearance 10.Īlthough the mechanism of Hsp70 function has been extensively studied, certain aspects of its structure-function features are yet to be fully established. Consequently, it was reasonable to expect that the involvement of specific Hsp70 isoforms could also contribute to dictate the final fate of client proteins. It is important to note that whereas the prokaryotic system has a single Hsp70 (the archetypical DnaK), eukaryotes have an expanded number of genes that code for distinct Hsp70 isoforms, which function in different physiological conditions and subcellular locations 2. Whereas classic client proteins bind to the Hsp70 SBD base domain, the KFERQ-containing proteins bind to a distinct, not yet determined, binding site 12. The color code of dots in interacting proteins indicates their Hsp70-binding domain-green: nucleotide-binding domain (NBD) red: substrate-binding domain (SBD) blue: C-terminal domain (CTD). The fate of a given Hsp70-bound protein (client proteins containing the classic hydrophobic motif or KFERQ-containing proteins) is dictated by the combination of Hsp70 interactors in a given cellular context. “Multiple socket” model for heat shock protein 70 (Hsp70) function in metazoan cells. The final combination of interactors dictates the fate of the Hsp70 client proteins 2 ( Figure 1).įigure 1. 
Alternatively, we proposed that Hsp70 could be playing its master role in protein homeostasis in a more passive way, namely as a “multiple socket” that provides a physical platform for the binding of client proteins and for the interaction with different chaperones and cochaperones. Cochaperone carboxyl terminus of Hsp70-interacting protein (CHIP) and Bag1 cochaperones have also been given this triage attribute independently 4– 7. Hsp70 has been referred to as the triage chaperone 3 as it determines the fate of client proteins. Hsp70 provides specificity for substrate selection to all of these degradation pathways in certain cellular contexts 2. There are four major protein degradation pathways in mammalian cells: the ubiquitin-proteasome system (UPS) and three types of autophagy: macroautophagy, microautophagy, and chaperone-mediated autophagy (CMA). 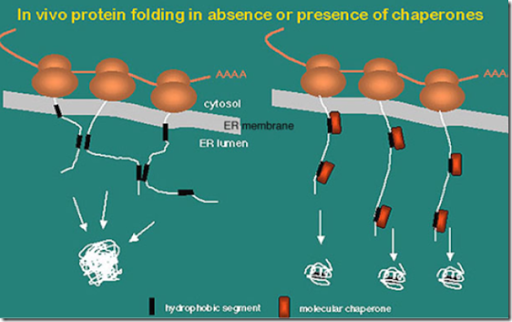
Molecular chaperones are important players for proteostasis in particular, the chaperone heat shock protein 70 (Hsp70) has an essential role in protein folding, disaggregation, and degradation 1. Impaired proteostasis occurs in aging and is associated with several human diseases. Protein homeostasis (proteostasis)-the balance of protein synthesis, folding, trafficking, assembly, and degradation-is essential for correct cellular function, and cells have developed a number of strategies to control it under stress. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
